Appropriateness scores according to expert panel
1. Each panellist independently performs appropriateness ratings
- Ratings from a clinical/scientific perspective
- Advice from the expert to a non-specialised colleague
- Treatment in daily clinical practice, not within a trial
- Not taken into consideration: financial costs, reimbursement, off-label use
2. Panel outcome is assessed using the rules of the RAND/UCLA appropriateness method
- Disagreement: at least 1/3 of the panellists rated in each extreme segment
- In case of disagreement, the panel outcome is uncertain by definition
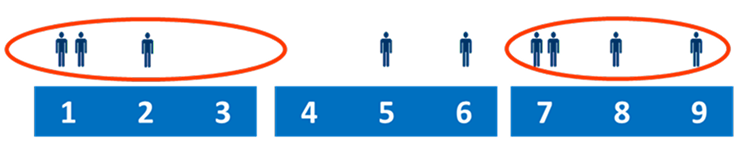
Panel outcomes
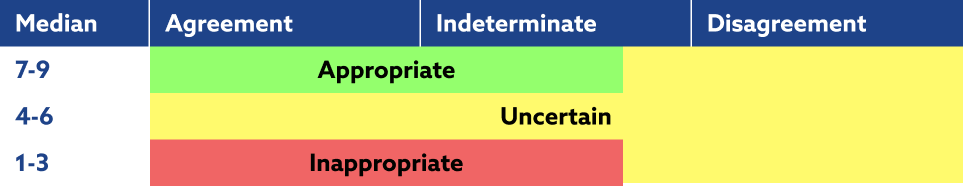
1-3
Inappropriate:Not recommended in this patient
4-6
Uncertain:No strong arguments in favour or against this option in this patient
7-9
Appropriate:Recommended in this patient
Expected benefits outweigh the expected risks/negative consequences by a sufficient margin